Industrializing biomaterials: Five critical transitions in scale-up
Authors: Tuija Vartia, Jarno Peltonen
The biomaterials sector has rarely been better positioned. Despite economic recession, investment plans flourish, policy frameworks are strengthening, and the pipeline of promising technologies has never been deeper. Still, moving toward a commercial facility remains difficult, and the challenges are often unexpected. In this article, biomaterials experts Tuija Vartia and Jarno Peltonen discuss why biomaterial innovations often fail to scale from laboratory to industrial production.
Walk the floor of any biomaterials industry event and you will find no shortage of ambition. What you find less often is an honest conversation about why so many biomaterial ventures that clear the scientific hurdles often struggle to scale to industrial production.
The failure rate at the transition from laboratory to commercial scale is high, and it is not primarily driven by bad science. It is driven by compounding challenges that are – based on Elomatic’s experience with startups – partly technical, partly organizational, partly financial, and partly commercial. Understanding where those challenges cluster, and what separates the ventures that navigate them from those that don’t, is the subject of this article.
A Journey that looks linear – but isn’t
Typical biomaterials industrialization process has distinct phases: ideation, laboratory development, pilot and/or demonstration plant, commercial operation, and replication. Presented as a diagram, this progression looks orderly and manageable. In practice, each transition represents a different risk profile and a different set of things that can go wrong. The costliest mistake is assuming that success in one phase reliably predicts success in the next.
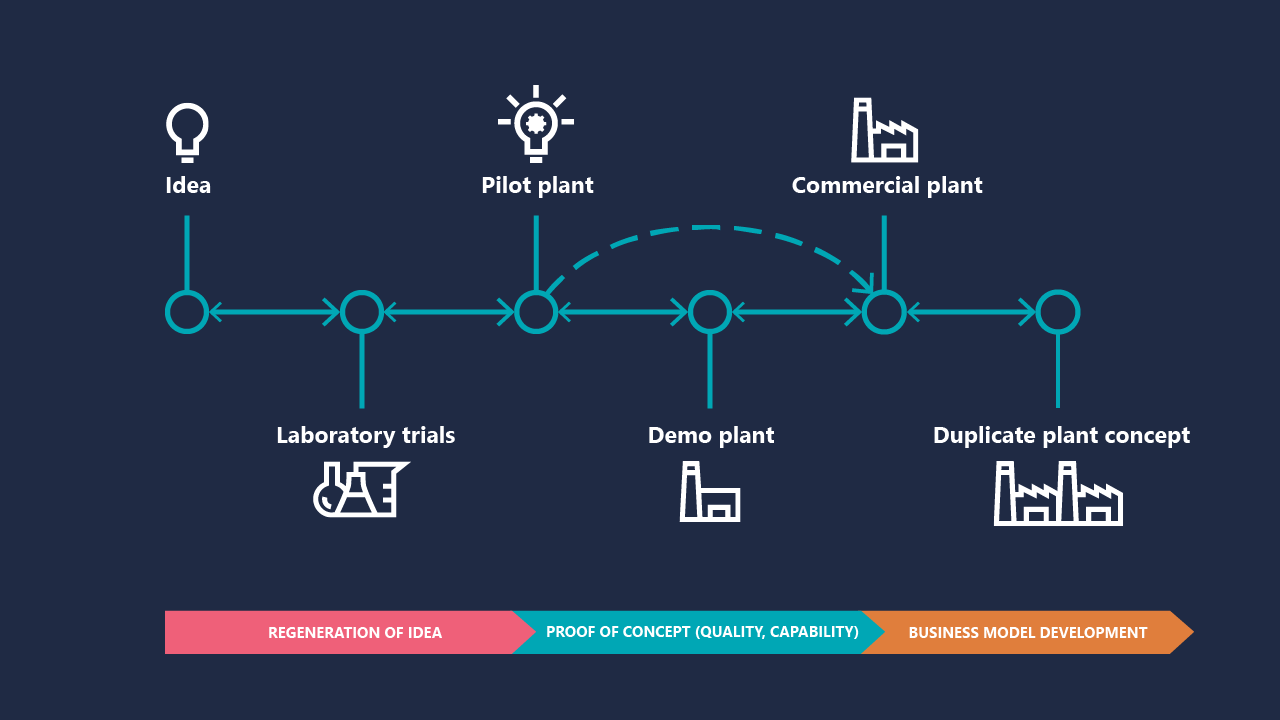
The ideation-to-laboratory transition is where foundational assumptions are made, and their quality will determine how much pain comes later. The ventures that fare best at this stage are those that design their early experimental work to stress-test assumptions rather than to confirm them. There is a meaningful difference between asking “does this process work under optimal conditions?” and asking “what conditions does this process need, and how realistic is it to provide them at industrial scale?”
Asking the industrial questions early, even when the answers might be uncomfortable, is one of the major differences between ventures that reach commercial scale and those that stall – because most often something can be done to those inconvenient answers.
The laboratory-to-pilot gap is bigger than it looks
The laboratory-to-pilot transition is where bio-based processes reveal their industrial character, and where real engineering constraints emerge.
As Holland et al. note in Engineering Biology, bio-based feedstock is a prime example. Laboratory work uses clean, consistent inputs. Industrial feedstock is chemically complex, with availability and quality varying by season, location, and supplier. Processes that work elegantly at litre scale become engineering challenges at cubic metre scale. Yields robust in batch experiments prove sensitive to the continuous operation that industrial economics demand.
Managing feedstock variability and the inherent variability of bio-based processes requires fundamentally different skills than laboratory work. This is where the capability gap becomes apparent: designing, commissioning, and operating a pilot plant demands expertise genuinely different from conducting research. In our experience, this gap is often underestimated in industrialization.
The most common failure mode isn’t technical
Ask most people what causes ventures to fail at scale-up, and the answers cluster around technology risk, capital, and market timing. These are real factors. But capability mismatch appears at least as frequently and is discussed far less openly.
Even the academia has relatively recently started to tackle the matter, as discussed by Szathmári et al. in Frontiers in Psychology. The founding teams of biomaterial ventures are typically strong where the venture began — in the science — and often weaker in the disciplines that industrial scale-up requires: plant operations management, procurement and supply chain development, regulatory affairs, and the commercial skills needed to negotiate offtake agreements and financing structures.
The ventures that manage this transition well treat organizational design as a strategic priority, thinking early about the capability architecture achieving their commercial-scale operation will require, and building toward it through hiring, advisory relationships, and industrial partnerships well before the demonstration plant is commissioned.
The hardest transition: commercial scale
If there is a single point where ventures are most vulnerable, it is the move from pilot/demonstration to commercial scale, in which every challenge arrives simultaneously. Capital requirements make a quantum leap. Technical risk, while reduced, has rarely fully resolved.
Regulatory complexity also increases. And the customer must now make a real procurement decision with real switching costs. Kampers et al discuss in Trends in Biotechnology the brevity of the Valley of Death and means to narrow it, but eventually, successfully crossing it requires a proof of commercially viable concept.
This is what makes the first-of-a-kind (FOAK) commercial plant such a distinctive challenge. It is simultaneously a production asset, a risk-resolution exercise, a regulatory process, and a commercial proof point.
Venture capital and regulatory constraints in first-of-a-kind projects
Based on our experience, traditional venture capital moves too fast for first-of-a-kind projects. The capital these projects need is patient and structured: combinations of strategic industrial investment, public financing, and offtake-backed debt.
As Gatto et al. note in Sustainability, assembling the financial architecture requires sophistication, lead time, and sustainability built into the business model from the start, not added later.
Regulation and sustainability add their own layer of complexity. Permitting timelines are rarely short, LCA methodologies are evolving inconsistently across standards bodies, and certification schemes have proliferated in ways that create both opportunity and confusion.
Typically, the ventures that manage this all most effectively treat regulatory and sustainability strategy as a design input from the outset. The reward for doing so is significant: in a growing number of markets, sustainability credentials are becoming prerequisites for access rather than differentiators within it.
The demand side has its own scale-up problem
Scale-up conversations focus almost entirely on supply. The demand side receives less attention, but it can kill a venture just as easily.
Large industrial buying processes aren’t configured for novel suppliers. Specification changes require lengthy approvals, supply risk frameworks expect commodity reliability, and price premiums need business cases that can’t be built without customers. Meanwhile, without binding offtakes, securing project financing becomes nearly impossible.
Market development must therefore start years before commercial volumes exist. Successful ventures invest early in deep customer relationships through pilot programs, co-development partnerships, and joint specification work that build buyer readiness alongside production readiness.
A well-structured long-term offtake agreement provides more than revenue: it enables project financing and signals market validation to investors.
Securing offtake agreements requires commercial sophistication, but it’s among the highest-return investments a pre-commercial venture can make.
How to build ventures that last?
Amid all these challenges, the ventures that succeed share five defining characteristics.
- Industrial reality: The ventures that last build interdisciplinary teams with genuine industrial depth early: not as support functions, but as strategic capabilities alongside the science.
- Strong partnerships: They develop value chain partnerships that distribute risk and accelerate learning across feedstock suppliers, technology partners, and anchor customers.
- The right capital at the right time: They secure capital structured for realistic timelines, understanding that mismatched capital – patient technology with impatient money – is a structural problem no technical performance can resolve.
- Compliance as an asset: They integrate regulatory and sustainability strategy as a design input already from ideation, not as an afterthought.
- Understanding the scale-up requirements: Most fundamentally, they treat scale-up as a first-class discipline in its own right, with the organizational architecture, the budget, and the leadership attention that discipline deserves.
In multiple scaleup projects, the companies that build durable industrial positions in biomaterials are not always those with the most innovative science. They are the ones that understand the full journey, prepare for each transition deliberately, and build the human and organizational architecture to match their technical ambition.
Sources:
Gatto, F., & Re, I. (2021). Circular Bioeconomy Business Models to Overcome the Valley of Death: A Systematic Statistical Analysis of Studies and Projects in Emerging Bio-Based Technologies and Trends Linked to the SME Instrument Support. Sustainability, 13(4), 1899.
Holland, C. & Shapira, P. Building the Bioeconomy: A Targeted Assessment Approach to Identifying Biobased Technologies, Challenges and Opportunities”. Eng. Biol. (2024):8, 1-15.
Kampers, L.F.C., Asín-García, E., Schaap, P.J., Wagemakers, A., & Martins dos Santos, V.A.P. (2021). From Innovation to Application: Bridging the Valley of Death in Industrial Biotechnology. Trends in Biotechnology, 39(12), 1240–1242.
Szathmári, E., Varga, Z., Molnár, A., Németh, G., Szabó, Z.P., & Kiss, O.E. (2024). Why Do Startups Fail? A Core Competency Deficit Model. Frontiers in Psychology, 15, 1299135.
Contact us

Read more:
World’s first bio-based acoustic panels to be manufactured soon at Aisti’s factory – Elomatic as key partner in plant implementation
Finnish startup Aisti has developed a bio-based, recyclable acoustic panel whose popularity surprised even the company’s founders. Elomatic has served as the company’s consulting and design partner since the pilot plant phase and is now helping Aisti bring the first products to market. The innovative product has required creative problem-solving in production line engineering: when suitable solutions were not available on the market, they were designed in-house.
New fiber from textile waste – Elomatic helps Infinited Fiber Company in their important mission
Infinited Fiber Company’s technology is globally unique. Thanks to it, textile waste can be transformed into high-quality fiber for the use in the textile industry. Elomatic has played a key role in building and developing the pilot production, and now the companies will continue their cooperation within Infinited Fiber Company’s factory project in Kemi.